December 19, 2022
The visualization of dopamine neurons
Discovery of region-specific imprinted expression of key factors involved in dopamine production in the brain
A study group led by Dr. Shinpei Yamaguchi at the Department of Biology, Faculty of Science, Toho University, utilized genome editing technology and developmental engineering techniques to develop a fluorescent reporter mouse model that can visualize dopamine neurons. This model enables the determination of whether the Dopa decarboxylase (Ddc, also known as Aadc) gene, an essential gene for dopamine production, is expressed on the chromosomes of paternal or maternal origin at the single-cell level. This mouse model will not only provide a foundation for elucidating the distinct functions of paternal- and maternal-derived chromosomes in behavior and other higher-order behaviors but will also be used for the etiological analysis of Parkinson’s disease and other diseases. The study was conducted in collaboration with Dr. Toru Nakano and his colleagues at Osaka University and published on December 7, 2022, in Frontiers in Cell and Developmental Biology.

The phenomenon of genomic imprinting, in which genes are expressed only from paternal or maternal alleles, is critical in normal mammalian development. Dopa decarboxylase is an enzyme that catalyzes the conversion of L-dopa to dopamine, which is encoded by the Ddc gene. The pattern of Ddc expression in the brain remains unclear. To examine the expression of Ddc, we generated red fluorescent protein knock-in reporter (Ddc-hKO1) mice. The detailed analysis of the brains of heterozygous mice carrying the Ddc-hKO1 gene on either the paternal or maternal chromosome revealed that Ddc is preferentially expressed from the maternal chromosome in the major dopamine neurons in the midbrain, from both chromosomes in neurons located in the midbrain to the brainstem, and from the paternal chromosome in the hypothalamus. In neurons located from the midbrain to the brainstem, expression is preferentially derived from both chromosomes. Each Ddc-expressing neuron has a unique function in its domain, such as feeding, motivation, and learning. It is believed that paternally and maternally derived chromosomes function differently in these neurons, which could help with aging and disease research.

The phenomenon of genomic imprinting, in which genes are expressed only from paternal or maternal alleles, is critical in normal mammalian development. Dopa decarboxylase is an enzyme that catalyzes the conversion of L-dopa to dopamine, which is encoded by the Ddc gene. The pattern of Ddc expression in the brain remains unclear. To examine the expression of Ddc, we generated red fluorescent protein knock-in reporter (Ddc-hKO1) mice. The detailed analysis of the brains of heterozygous mice carrying the Ddc-hKO1 gene on either the paternal or maternal chromosome revealed that Ddc is preferentially expressed from the maternal chromosome in the major dopamine neurons in the midbrain, from both chromosomes in neurons located in the midbrain to the brainstem, and from the paternal chromosome in the hypothalamus. In neurons located from the midbrain to the brainstem, expression is preferentially derived from both chromosomes. Each Ddc-expressing neuron has a unique function in its domain, such as feeding, motivation, and learning. It is believed that paternally and maternally derived chromosomes function differently in these neurons, which could help with aging and disease research.
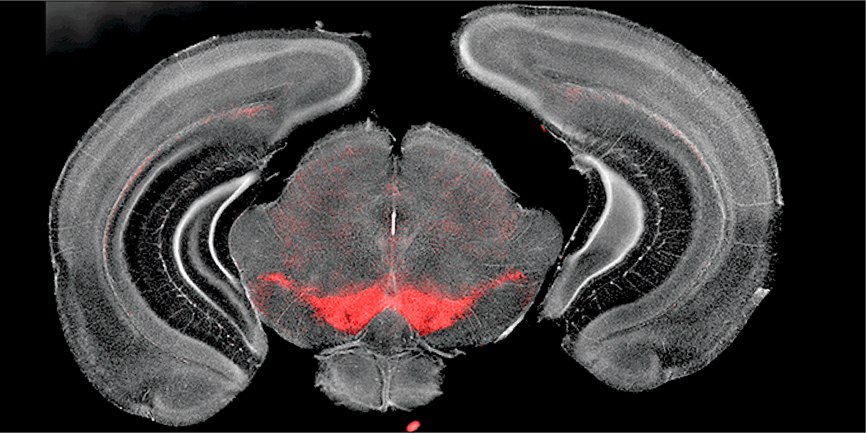
Figure 1: Representative images of the coronal plane section of adult Ddc-hKO1 homozygous mice. Ddc-hKO1 signal was detected at VTA and SNc regions
Journal:
Frontiers in Cell and Developmental Biology December 7, 2022 issue
Title:
A region-dependent allele-biased expression of Dopa decarboxylase in mouse brain
Authors:
Kit-Yeng Sheng, Toru Nakano, and Shinpei Yamaguchi*
DOI No.
https://doi.org/10.3389/fcell.2022.1078927
Frontiers in Cell and Developmental Biology December 7, 2022 issue
Title:
A region-dependent allele-biased expression of Dopa decarboxylase in mouse brain
Authors:
Kit-Yeng Sheng, Toru Nakano, and Shinpei Yamaguchi*
DOI No.
https://doi.org/10.3389/fcell.2022.1078927
READ MORE RESEARCH NEWS - SCIENCE
ACADEMICS
Undergraduate Programs
– Medicine
– Pharmaceutical Sciences
– Science
– Nursing
– Health Science
Graduate Programs
–Medicine
–Pharmaceutical Sciences
–Science
–Nursing
Undergraduate Programs
– Medicine
– Pharmaceutical Sciences
– Science
– Nursing
– Health Science
Graduate Programs
–Medicine
–Pharmaceutical Sciences
–Science
–Nursing
RESEARCH
– News
– Guidelines & Policies
– Support Offices
– Facilities
– Security Export Control
Non-Degree Programs
– Clinical Elective Program
– International Physician Observership Program